The Vaccine Manufacturing and Quality Assurance Architecture
A Business Architecture View for Closed Loop Process Manufacturing and Closed Loop of Quality
- Vaccine Manufacturing is a biological process that requires high level of expertise and control systems leverage. The entire process takes on an average 7-36 months to produce, package and deliver a vaccine. Each batch thereafter is released at every 45-60 days.
- Process is the focus based on the Good Practices related to Non-Clinical during R&D (GLP), Clinical (GCP), Manufacturing (GMP), and Distribution (GDP) the focus is on quality (70% of time is spent on quality control) and the stringent adherence to process and outcomes traceability.
- Outcomes dependent on the stringency of process, and controls for management of “specification limits” for storage, logistics, biosafety guideline, and execution standard operating procedures. Any variations to these specification limits directly impact the safety, efficacy, purity and potency of the vaccine.
- “Thus, digitization post marketing authorization phase of vaccine manufacturing expects technologies and Industrial IoT for integrating distributed control systems with IT (ERP) and Operational Technologies (MES, SCADA, Cold Chain Sensors and Controls) for ensuring expected outcomes compliant to the Regulatory Framework. The Closed Loop Manufacturing and Quality Architecture sets the foundation for its operationalization”.
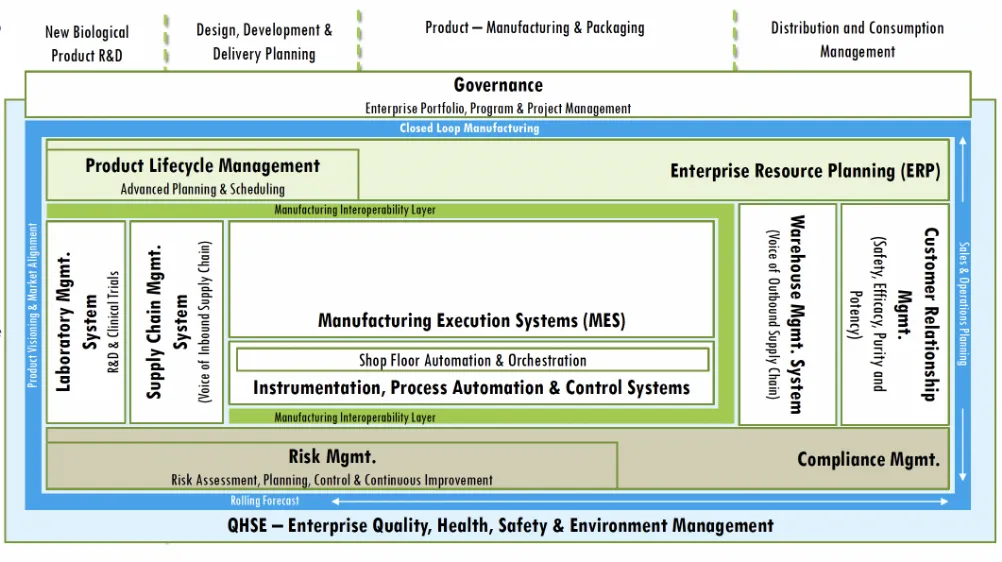
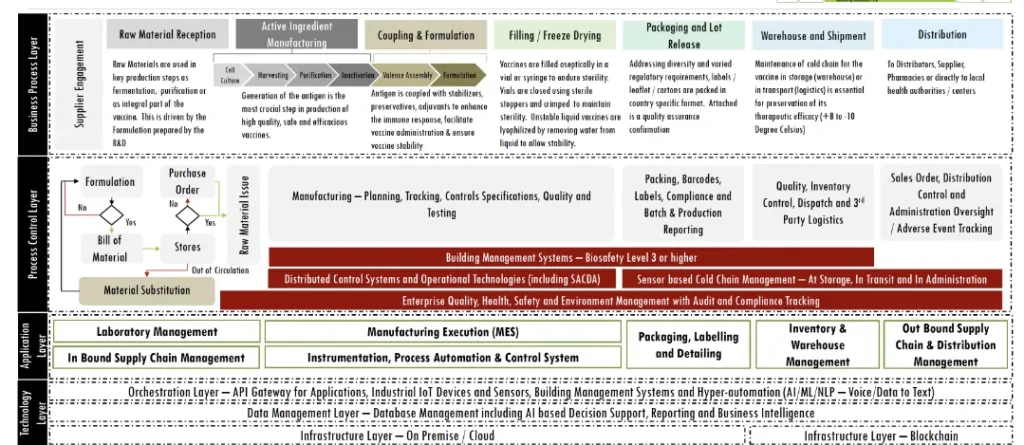
- Design for Compliance: Proposed Business Process Model is based on the stringent Instrumentation and Process Control Frameworks for enabling faster Go To Market Authorizations and focus on Safety, Efficacy, Purity and Potency as per the Regulator’s applicable guidelines integrated with Digital Signatures, IP, OP, and PQ
- Design for Automation: Being a “rules driven” and “predictable process model” based approach, the approach is made amenable to hyper automation with BoTs as eWorkers for repetitive and low value-added tasks with Voice / Written to Text conversion leveraging NLP
- Design for Insights: Based on a foundation for entity data mapping, the solution will deliver insights for Decision Support & Compliance Authorization
- Designed for Interoperability: Leveraging Blockchain vaccine antecedent tracking while remaining immutable, tamper proof, trusted across heterogenous data system
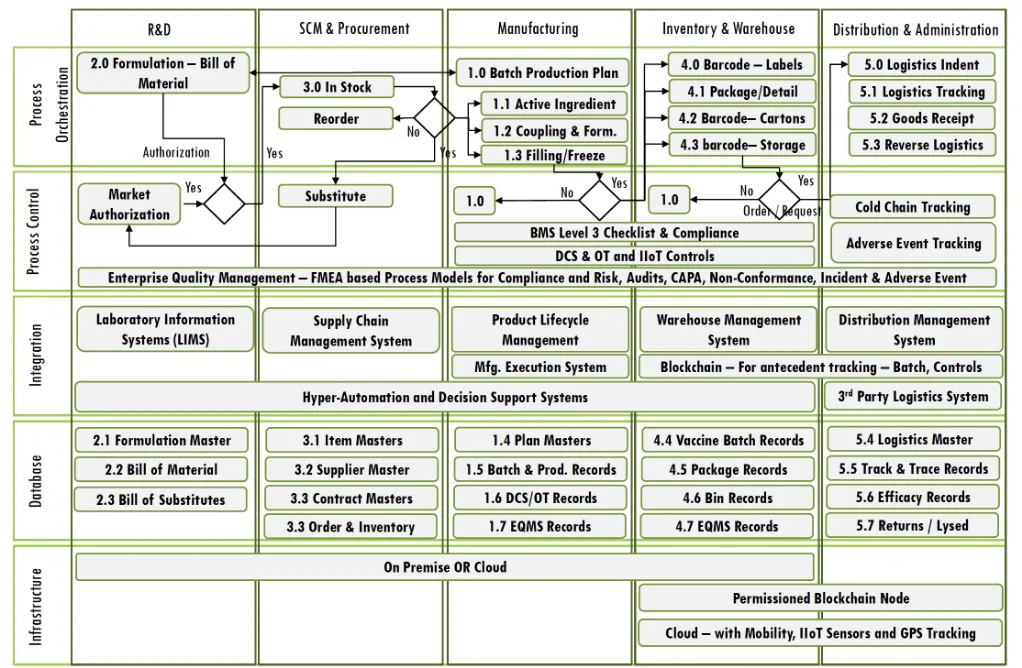
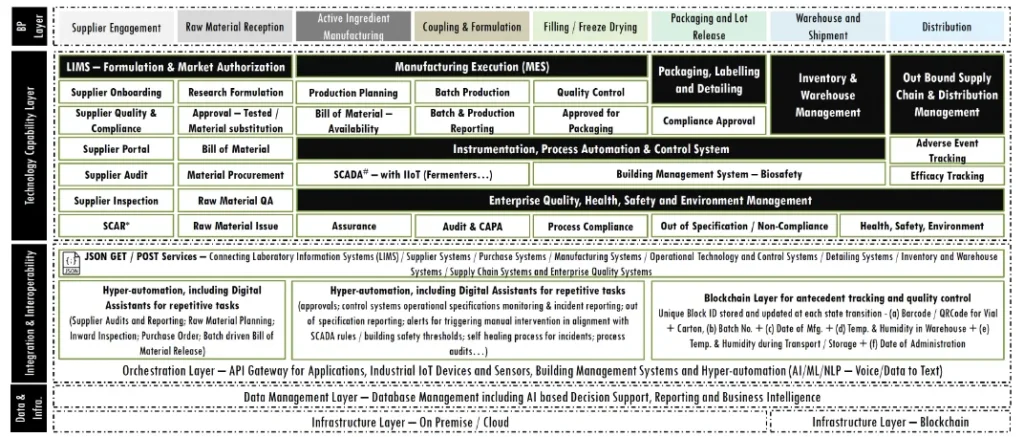
Application Landscape proposed,
- Laboratory Information Management including Formulation, Approval, Bill of Material and Raw Material Issue
- Supplier and Purchase Management including Supplier Management, Purchase Order to GRN Tracking and Account Payables
- Production Planning and Execution Tracking and Management
- Packaging, Labelling and Detailing
- Inventory and Warehouse Management
- SCADA Systems for Instrumentation and Process Engineering including integrations with the Devices and Sensors
- Enterprise Quality Management Systems for quality and compliance management including 21CFR Part 11
tracking - 3rd Party Logistics Track and Trace, including cold chain sensor integrations planned for XX transportation units
- Hyper automation for AI driven RPA with an aim for 10 processes to be automated in the Year 1 and scaling to 15 by year 2 and beyond using a full stack AI Platform
- Integration aims to leverage the Service Bus and API gateway
Instrumentation & Process Control proposed,
- It was assumed that all the instruments and devices used will be IoT based with sensing and open API based sharing of device control data and the actual functional data
- A supervisory control and data acquisition (SCADA) platform was planned for integrated management of the various process control system comprising computers, devices and networked data communications with graphical user interfaces (GUI) for high level process supervisory management
Outcomes
This case study demonstrates how Enterprise Architecture—when applied as a business capability enabler rather than an IT blueprint—can materially compress time‑to‑market, improve regulatory confidence, and create a scalable foundation for biologics manufacturing in highly regulated environments.
| Dimension | Enterprise Architecture Impact |
|---|---|
| Time to Market | Parallel quality & production, API‑led integration, faster batch release |
| Automation | Embedded hyper‑automation, event‑driven workflows, compliance automation |
| Process Optimization | Closed‑loop manufacturing & quality, standardized process models |
| Compliance & Risk | Immutable blockchain traceability, audit‑ready processes |
| Scalability | Cloud‑native, loosely coupled, partner‑friendly architecture |
© 2026 Vivikta Advisory. All rights reserved. The views, opinions, and analyses expressed in this content represent those of Vivikta Advisory and its employees only.
No part of this content may be quoted, reproduced, distributed, or used in any form without prior written permission from Vivikta Advisory.
This material is created for informational purposes and does not intentionally reference, resemble, or imply any specific persons, organizations, or proprietary information. Vivikta Advisory disclaims any liability arising from reliance on this content.
